.png)
What is Clinion CTMS?
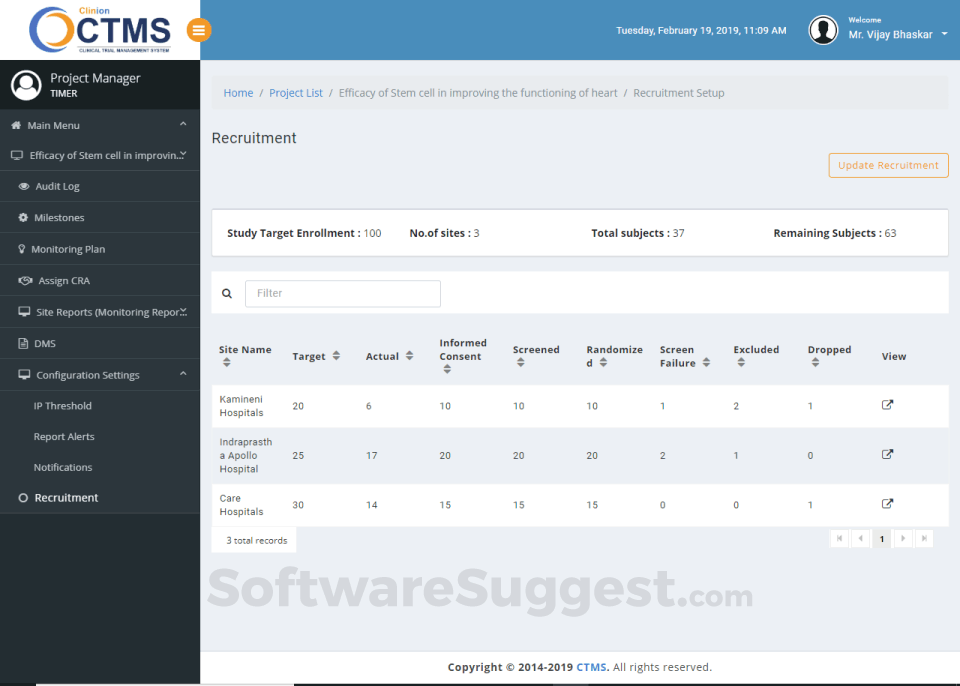
Clinion CTMS is a Clinical Trial Management Software designed to serve Enterprises, Agencies. Clinion CTMS provides end-to-end solutions designed for Web App. This online Clinical Trial Management system offers 21 CFR Part 11 Compliance, Document Management, Monitoring, Scheduling, Study Planning one place.
Clinion CTMS Starting Price

Our Awards and Recognition
Talk with a software expert for free. Get a list of software that's great for you in less than 10 minutes.
Key Features of Clinion CTMS
Here are the powerful features that make Clinion CTMS stand out from the competition. Packed with innovative tools and advanced functionalities, this software offers a range of benefits.
- 21 CFR Part 11 Compliance
- Analytics
- Audience Targeting
- Audit Trail
- Campaign Management
- Clinical Communication and Collaboration
- Clinical Trial Management
- Comparison View
- Compliance Management
- Content Syndication
- Conversion Tracking
- CRF Tracking
- Dashboard
- Data Analysis Auditing
- Data Entry
- Data Import / Export
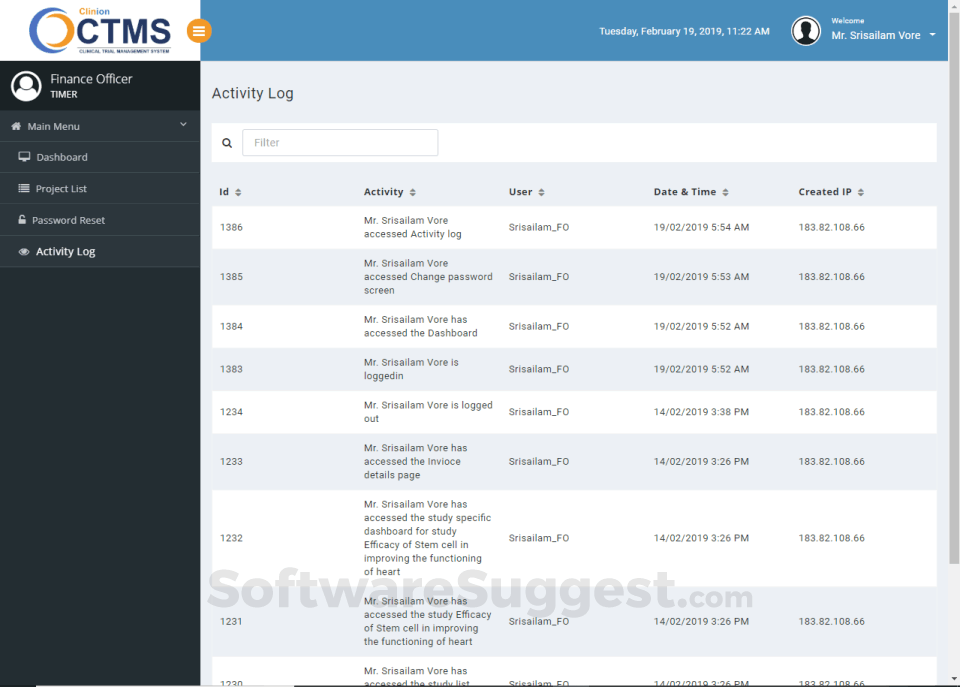
- Audit trail and data tracking
- Automated data validation and edit checks
- Centralized data management
- Data anonymization and de-identification
- Data cleaning and validation checks
- Data encryption and security
- Data entry and validation
- Data export for regulatory submissions
- Data export in multiple formats (CSV
- Excel
- etc.)
- Data export to statistical analysis tools
- Data Import and Export
- Data integration with other systems
- Data monitoring and quality control
- Data query management
Clinion CTMS Specifications
Get a closer look at the technical specifications and system requirements for Clinion CTMS. Find out if it's compatible with your operating system and other software.
Clinion CTMS Description
Here's the comprehensive description of Clinion CTMS. Gain a brief understanding of its unique features and exceptional benefits.
Clinion CTMS
Most Easy-to-Use Clinical Trial Management System (CTMS)
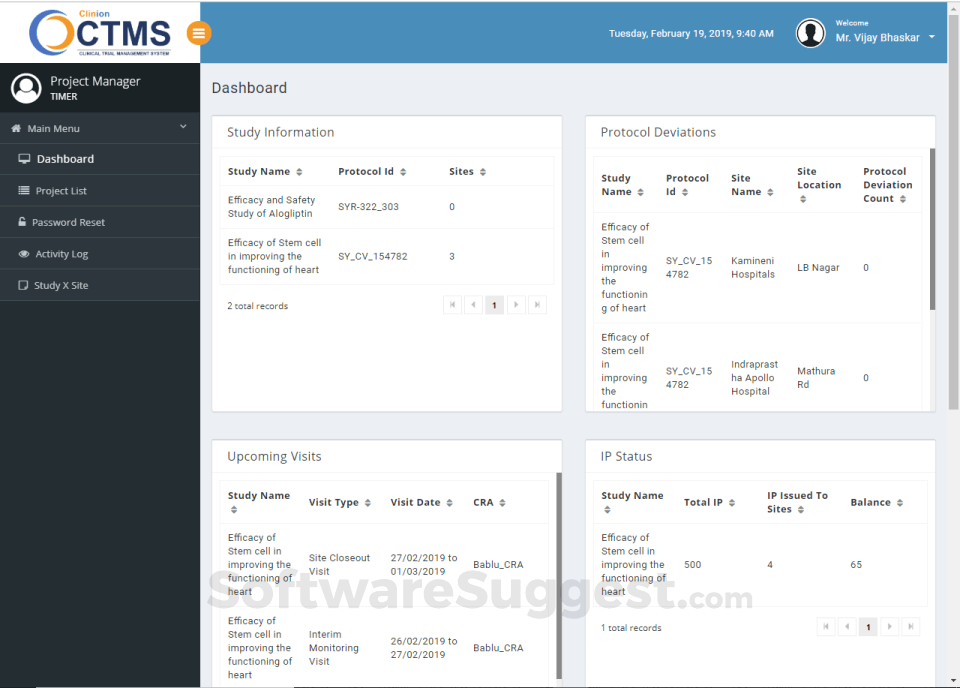
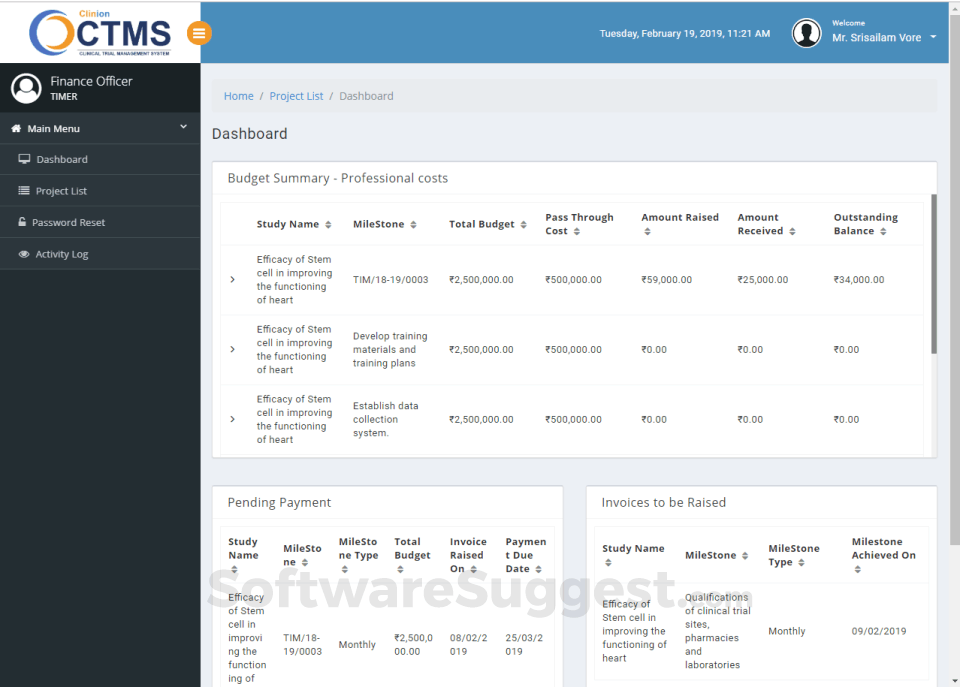
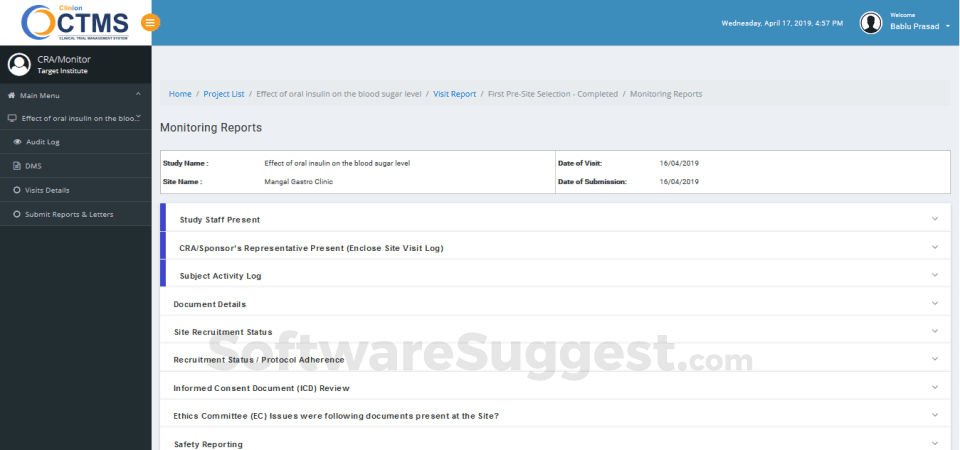
Integrates seamlessly with Clinion EDC and Clinion RTSM to provide detailed and faster access to the entire trial process to address all aspects of Clinical trial management from a single platform: investigator sites, patients enrollment, SAEs, deviations, inclusions, visit scheduling, IP management, milestones, finances, custom reports, and on-demand analytics enables you to improve quality, control risk, and reduce costs.
Our Clinical Trial Management System allows CROs and Sponsors to monitor the trial progress actively with its advanced dashboard and alert notification system. So you’ll never miss a deadline again!
What can you expect?
- Comprehensive CTMS Software
- Single Source for your entire trial data
- On-demand Analytics for Risk-Based Management of Clinical Trials
- Fully Integrated with Clinion EDC and Clinion RTSM
- 100% web-based and hosted securely on the cloud
- 21 CFR Part 11 & GDPR compliant
Clinion CTMS Resources

Clinion CTMS Videos
Clinion CTMS Reviews (0)
No reviews yet. Rate this app or be the first to review.
Write a ReviewAlternatives of Clinion CTMS
Explore alternative software options that can fulfill similar requirements as Clinion CTMS. Evaluate their features, pricing, and user feedback to find the perfect fit for your needs.
Clinion CTMS FAQs
What are the top 5 features for Clinion CTMS?
The top 5 features for Clinion CTMS are:
- Dashboard
- Analytics
- Scheduling
- Monitoring
- 21 CFR Part 11 Compliance
What type of customer support is available from Clinion CTMS?
The available support which Clinion CTMS provides is:
- Phone
- Live support
- Training
Where is the headquarters/company of Clinion CTMS located?
The headquarters/company of Clinion CTMS is located at Austin.